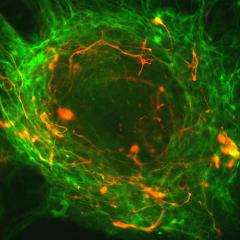
There is growing awareness that the long-standing unmet need for specific counting of therapeutic tissue stem cells has been and continues to be a significant impediment to progress in stem cell therapy, gene and gene editing therapies (because targeting tissue stem cells is essential for durable one-time treatments), and drug development (because...





